ALHABIB DENTAL CLINIC
ROOTCANAL TREATMENT
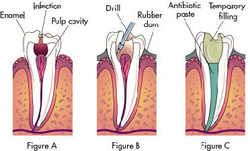
Endodontic therapy is a sequence of treatment for the pulp of a tooth which results in the elimination of infection and protection of the decontaminated tooth from future microbial invasion. This set of procedures is commonly referred to as a "root canal." Root canals and their associated pulp chamber are the physical hollows within a tooth that are naturally inhabited by nerve tissue, blood vessels and other cellular entities. Endodontic therapy involves the removal of these structures, the subsequent shaping, cleaning, and decontamination of the hollows with tiny files and irrigating solutions, and the obturation (filling) of the decontaminated canals with an inert filling such as gutta percha and typically a eugenol-based cement.
After endodontic surgery the tooth will be "dead," and if an infection is spread at apex, root end surgery is required.
Although the procedure is relatively painless when done properly[1], the root canal remains a stereotypically fearsome dental operation.
Contents
The standard filling material is gutta-percha, a natural non-elastic latex from the sap of the percha (Palaquium gutta) tree. The standard endodontic technique involves inserting a gutta-percha cone (a "point") into the cleaned-out root canal along with cement and a sealer.[2] Another technique uses melted or heat-softened gutta-percha which is then injected or pressed into the root canal passage(s). However, as gutta-percha shrinks as it cools, thermal techniques can be unreliable and sometimes a combination of techniques is used. Gutta-percha is http://root canal, alhabib dental clinic, teeth, tooth, camera, implant, allowing verification afterwards that the root canal passages have been completely filled in, without voids.
An alternative filling material was invented in the early 1950s by Angelo Sargenti. It has undergone several formulations over the years (N2, N2 Universal, RC-2B, RC-2B White), but all contain paraformaldehyde. The paraformaldehyde, when placed into the root canal, forms formaldehyde, which penetrates and sterilizes the passage. The formaldehyde is then theoretically transformed to harmless water and carbon dioxide. The outcome is better than a root canal done with gutta percha according to some investigations. There is however a lack of indisputable, scientifically made studies according to the Swedish Council on Health Technology Assessment.
In rare cases, the paste like any other material can be forced past the root tip into the surrounding bone. If this happens, the formaldehyde will immediately be transformed into a harmless substance. The blood normally contains 2 mg formaldehyde per liter and the body regulates this in seconds. The rest of an overfill will be gradually absorbed and the end result is normally good. In 1991 the ADA Council on Dental Therapeutics resolved that the treatment was "not recommended", and it is not taught in any American dental school. Scientific evidence in endodontic therapy was, and still is lacking.[3] The Sargenti technique has its advocates, however, who believe N2 to be less expensive and at least as safe as gutta-percha.[4]
For some patients, root canal therapy is one of the most feared dental procedures, perhaps because of a painful abscess that necessitated the root canal procedure. However, dental professionals assert that modern root canal treatment is relatively painless because the pain can be controlled with a local anesthetic during the procedure and pain control medication can be used before and/or after treatment assuming that the dentist takes the time to administer one. However, in some cases it may be very difficult to achieve pain control before performing a root canal. For example, if a patient has an abscessed tooth, with a swollen area or "fluid-filled gum blister" next to the tooth, the pus in the abscess may contain acids that inactivate any anesthetic injected around the tooth. In this case, the dentist may drain the abscess by cutting it to let the pus drain out. Releasing the pus releases pressure built up around the tooth; this pressure causes the pain. The dentist then prescribes a week of antibiotics such as penicillin, which will reduce the infection and pus, making it easier to anesthetize the tooth when the patient returns one week later. The dentist could also open up the tooth and let the pus drain through the tooth, and could leave the tooth open for a few days to help relieve pressure.
At this first visit, the dentist must ensure that the patient is not biting into the tooth, which could also trigger pain. Sometimes the dentist performs preliminary treatment of the tooth by removing all of the infected pulp of the tooth and applying a dressing and temporary filling to the tooth. This is called a pulpectomy. The dentist may also remove just the coronal portion of the dental pulp, which contains 90% of the nerve tissue, and leave intact the pulp in the canals. This procedure, called a "pulpotomy", tends to essentially eliminate all the pain. A pulpotomy may be a relatively definitive treatment for infected primary teeth. The pulpectomy and pulpotomy procedures eliminate almost all pain until the follow-up visit for finishing the root canal. But if the pain returns, it means any of three things: the patient is biting into the tooth, there is still a significant amount of sensitive nerve material left in the tooth, or there is still more pus building up inside and around the infected tooth; all of these cause pain.
After removing as much of the internal pulp as possible, the root canals can be temporarily filled with calcium hydroxide paste. This strong alkaline base is left in for a week or more to disinfect and reduce inflammation in surrounding tissue.[5] Ibuprofen taken orally is commonly used before and/or after these procedures to reduce inflammation. The following substances are used as root canal irrigants during the root canal procedure:
Pulp tissue removed during endodontic therapy by a size 20 broach file. The procedure is often complicated, depending on circumstances, and may involve multiple visits over a period of weeks. The cost is typically high.
Alternatives The alternatives to root canal therapy include no treatment, tooth extraction, or 3Mix-MP procedure. Following tooth extraction, a single missing tooth can be replaced with a dental implant, fixed partial denture (commonly known as a bridge), or by a removable partial denture. There are risks to no treatment, such as pain, infection, and the possibility of worsening dental infection such that the tooth will be no longer restorable (root canal treatment will not be successful, often due to excessive loss of tooth structure). If extensive loss of tooth structure occurs, extraction will be the only treatment option.
In December 2010, a study was published demonstrating a new alternative to root canal therapy in treating infected tooth pulps, 3Mix-MP procedure, through the local application of an antibacterial drug mixture.[7] While previous studies had failed in similar experiments, this study succeeded by utilizing a unique vehicle for the antibiotics, propylene glycol, which has been shown to successfully penetrate and spread through dentinal tubules.[8] This is an important finding which seems to offer an alternative to root canal therapy other than tooth extraction.
Innovation In the last ten to twenty years, there have been great innovations in the art and science of root canal therapy. Dentists now must be educated on the current concepts in order to optimally perform a root canal. Root canal therapy has become more automated and can be performed faster, thanks to advances in automated mechanical instrumentation of teeth and more advanced root canal filling methods. Most root canal procedures are done in one dental visit, lasting around 1–2 hours. Dentists also possess newer technologies that allow more efficient, scientific measurements to be taken of the dimensions of the root canal that must be filled. Many dentists use dental loupes to perform root canals, and the consensus is that root canals performed using loupes or other forms of magnification are more likely to succeed than those performed without them. Although general dentists are becoming versed in these advanced technologies, they are still more likely to be used by specialist root canal doctors (known as endodontists).
Laser root canal procedures are a controversial innovation. Lasers may be fast but have not been shown to thoroughly disinfect the whole tooth,[9] and may cause damage.[10]
Procedural accidents Instruments may separate (break) during root canal treatment, meaning a small portion of the metal file used during the procedure is separated inside the tooth. The file segment may be left behind if an acceptable level of cleaning and shaping has already been completed and attempting to remove the segment would risk damage to the tooth. While potentially disconcerting to the patient, having metal inside of a tooth is relatively common, such as with metal posts, amalgam fillings, gold crowns, and porcelain fused to metal crowns. The occurrence of file separation is proportional to the narrowness, curvature, length, calcification and number of roots on the tooth being treated. Complications resulting from incompletely cleaned canals, due to blockage from the separated file, can be addressed with surgical root canal treatment. The occurrence of instrument separation is well documented.[11]
Success and prognosis Root canal treated teeth may fail to heal, for example if the dentist does not find, clean and fill all of the root canals within a tooth. On a maxillary molar, there is a more than 50% chance that the tooth has four canals instead of just three. But the fourth canal, often called a "mesio-buccal 2", tends to be very difficult to see and often requires special instruments and magnification in order to see it (most commonly found in first maxillary molars; studies have shown an average of 76% up to 96% of such teeth with the presence of an MB2 canal). This infected canal may cause a continued infection or "flare up" of the tooth. Any tooth may have more canals than expected, and these canals may be missed when the root canal is performed. Sometimes canals may be unusually shaped, making them impossible to clean and fill completely; some infected material may remain in the canal. Sometimes the canal filling does not fully extend to the apex of the tooth, or it does not fill the canal as densely as it should. Sometimes a tooth root may be perforated while the root canal is being treated, making it difficult to fill the tooth. The perforation may be filled with a root repair material, such as one derived from natural cement called MTA. A specialist can often re-treat failing root canals, and these teeth will then heal, often years after the initial root canal procedure.
However, the survival or functionality of the endodontically-treated tooth is often the most important aspect of endodontic treatment outcomes, rather than apical healing alone.[12] Recent studies indicate that substances commonly used to clean the root canal space incompletely sterilize the canal.[13] A properly restored tooth following root canal therapy yields long-term success rates near 97%. In a large scale Delta Dental Study of over 1.6 million patients who had root canal therapy, 97% had retained their teeth 8 years following the procedure, with most untoward events, such as re-treatment, apical surgery or extraction, occurring during the first 3 years after the initial endodontic treatment.[14] Endodontically treated teeth are prone to extraction mainly due to non-restorable carious destruction and to a lesser extent to endodontic-related reasons such as endodontic failure, vertical root fracture, or perforation (procedural error).[6]
Systemic issues An infected tooth may endanger other parts of the body. People with special vulnerabilities, such as prosthetic joint replacement or mitral valve prolapse, may need to take antibiotics to protect from infection spreading during dental procedures. Both endodontic therapy and tooth extraction can lead to subsequent jaw bone infection. The American Dental Association (ADA) asserts that any risks can be adequately controlled.
In the early 1900s, several researchers theorized that bacteria from teeth which had necrotic pulps or which had received endodontic treatment could cause chronic or local infection in areas distant from the tooth through the transfer of bacteria through the bloodstream. This was called the "focal infection theory", and it led some dentists to advocate dental extraction. In the 1930s, this theory was discredited, but the theory was recently revived by a book entitled Root Canal Cover-Up Exposed which used the early discredited research, and further complicated by epidemiological studies which found correlations between periodontal disease and heart disease, strokes, and preterm births. Bacteremia (bacteria in the bloodstream) can be caused by dental procedures, particularly after dental extractions, but endodontically treated teeth alone do not cause bacteremia or systemic disease.[
After endodontic surgery the tooth will be "dead," and if an infection is spread at apex, root end surgery is required.
Although the procedure is relatively painless when done properly[1], the root canal remains a stereotypically fearsome dental operation.
Contents
- 1 Root canal treatment
- 2 Innovation
- 3 Procedural accidents
- 4 Success and prognosis
- 5 Systemic issues
- 6 See also
- 7 References
- 8 External links
The standard filling material is gutta-percha, a natural non-elastic latex from the sap of the percha (Palaquium gutta) tree. The standard endodontic technique involves inserting a gutta-percha cone (a "point") into the cleaned-out root canal along with cement and a sealer.[2] Another technique uses melted or heat-softened gutta-percha which is then injected or pressed into the root canal passage(s). However, as gutta-percha shrinks as it cools, thermal techniques can be unreliable and sometimes a combination of techniques is used. Gutta-percha is http://root canal, alhabib dental clinic, teeth, tooth, camera, implant, allowing verification afterwards that the root canal passages have been completely filled in, without voids.
An alternative filling material was invented in the early 1950s by Angelo Sargenti. It has undergone several formulations over the years (N2, N2 Universal, RC-2B, RC-2B White), but all contain paraformaldehyde. The paraformaldehyde, when placed into the root canal, forms formaldehyde, which penetrates and sterilizes the passage. The formaldehyde is then theoretically transformed to harmless water and carbon dioxide. The outcome is better than a root canal done with gutta percha according to some investigations. There is however a lack of indisputable, scientifically made studies according to the Swedish Council on Health Technology Assessment.
In rare cases, the paste like any other material can be forced past the root tip into the surrounding bone. If this happens, the formaldehyde will immediately be transformed into a harmless substance. The blood normally contains 2 mg formaldehyde per liter and the body regulates this in seconds. The rest of an overfill will be gradually absorbed and the end result is normally good. In 1991 the ADA Council on Dental Therapeutics resolved that the treatment was "not recommended", and it is not taught in any American dental school. Scientific evidence in endodontic therapy was, and still is lacking.[3] The Sargenti technique has its advocates, however, who believe N2 to be less expensive and at least as safe as gutta-percha.[4]
For some patients, root canal therapy is one of the most feared dental procedures, perhaps because of a painful abscess that necessitated the root canal procedure. However, dental professionals assert that modern root canal treatment is relatively painless because the pain can be controlled with a local anesthetic during the procedure and pain control medication can be used before and/or after treatment assuming that the dentist takes the time to administer one. However, in some cases it may be very difficult to achieve pain control before performing a root canal. For example, if a patient has an abscessed tooth, with a swollen area or "fluid-filled gum blister" next to the tooth, the pus in the abscess may contain acids that inactivate any anesthetic injected around the tooth. In this case, the dentist may drain the abscess by cutting it to let the pus drain out. Releasing the pus releases pressure built up around the tooth; this pressure causes the pain. The dentist then prescribes a week of antibiotics such as penicillin, which will reduce the infection and pus, making it easier to anesthetize the tooth when the patient returns one week later. The dentist could also open up the tooth and let the pus drain through the tooth, and could leave the tooth open for a few days to help relieve pressure.
At this first visit, the dentist must ensure that the patient is not biting into the tooth, which could also trigger pain. Sometimes the dentist performs preliminary treatment of the tooth by removing all of the infected pulp of the tooth and applying a dressing and temporary filling to the tooth. This is called a pulpectomy. The dentist may also remove just the coronal portion of the dental pulp, which contains 90% of the nerve tissue, and leave intact the pulp in the canals. This procedure, called a "pulpotomy", tends to essentially eliminate all the pain. A pulpotomy may be a relatively definitive treatment for infected primary teeth. The pulpectomy and pulpotomy procedures eliminate almost all pain until the follow-up visit for finishing the root canal. But if the pain returns, it means any of three things: the patient is biting into the tooth, there is still a significant amount of sensitive nerve material left in the tooth, or there is still more pus building up inside and around the infected tooth; all of these cause pain.
After removing as much of the internal pulp as possible, the root canals can be temporarily filled with calcium hydroxide paste. This strong alkaline base is left in for a week or more to disinfect and reduce inflammation in surrounding tissue.[5] Ibuprofen taken orally is commonly used before and/or after these procedures to reduce inflammation. The following substances are used as root canal irrigants during the root canal procedure:
- 5.25% sodium hypochlorite (NaOCl)
- 6% sodium hypochlorite with surface modifiers for better flow into nooks and crannies
- 2% chlorhexidine gluconate (Perioxidina Plus-2)
- 0.2% chlorhexidine gluconate plus 0.2% cetrimide (Cetrexidin)
- 17% ethylenediaminetetraacetic acid (EDTA)
- Framycetin sulfate (Septomixine)
- Biopure MTAD Mixture of citric acid, Docycline, and Tween-80 (detergent) by Dentsply USA (MTAD)
Pulp tissue removed during endodontic therapy by a size 20 broach file. The procedure is often complicated, depending on circumstances, and may involve multiple visits over a period of weeks. The cost is typically high.
Alternatives The alternatives to root canal therapy include no treatment, tooth extraction, or 3Mix-MP procedure. Following tooth extraction, a single missing tooth can be replaced with a dental implant, fixed partial denture (commonly known as a bridge), or by a removable partial denture. There are risks to no treatment, such as pain, infection, and the possibility of worsening dental infection such that the tooth will be no longer restorable (root canal treatment will not be successful, often due to excessive loss of tooth structure). If extensive loss of tooth structure occurs, extraction will be the only treatment option.
In December 2010, a study was published demonstrating a new alternative to root canal therapy in treating infected tooth pulps, 3Mix-MP procedure, through the local application of an antibacterial drug mixture.[7] While previous studies had failed in similar experiments, this study succeeded by utilizing a unique vehicle for the antibiotics, propylene glycol, which has been shown to successfully penetrate and spread through dentinal tubules.[8] This is an important finding which seems to offer an alternative to root canal therapy other than tooth extraction.
Innovation In the last ten to twenty years, there have been great innovations in the art and science of root canal therapy. Dentists now must be educated on the current concepts in order to optimally perform a root canal. Root canal therapy has become more automated and can be performed faster, thanks to advances in automated mechanical instrumentation of teeth and more advanced root canal filling methods. Most root canal procedures are done in one dental visit, lasting around 1–2 hours. Dentists also possess newer technologies that allow more efficient, scientific measurements to be taken of the dimensions of the root canal that must be filled. Many dentists use dental loupes to perform root canals, and the consensus is that root canals performed using loupes or other forms of magnification are more likely to succeed than those performed without them. Although general dentists are becoming versed in these advanced technologies, they are still more likely to be used by specialist root canal doctors (known as endodontists).
Laser root canal procedures are a controversial innovation. Lasers may be fast but have not been shown to thoroughly disinfect the whole tooth,[9] and may cause damage.[10]
Procedural accidents Instruments may separate (break) during root canal treatment, meaning a small portion of the metal file used during the procedure is separated inside the tooth. The file segment may be left behind if an acceptable level of cleaning and shaping has already been completed and attempting to remove the segment would risk damage to the tooth. While potentially disconcerting to the patient, having metal inside of a tooth is relatively common, such as with metal posts, amalgam fillings, gold crowns, and porcelain fused to metal crowns. The occurrence of file separation is proportional to the narrowness, curvature, length, calcification and number of roots on the tooth being treated. Complications resulting from incompletely cleaned canals, due to blockage from the separated file, can be addressed with surgical root canal treatment. The occurrence of instrument separation is well documented.[11]
Success and prognosis Root canal treated teeth may fail to heal, for example if the dentist does not find, clean and fill all of the root canals within a tooth. On a maxillary molar, there is a more than 50% chance that the tooth has four canals instead of just three. But the fourth canal, often called a "mesio-buccal 2", tends to be very difficult to see and often requires special instruments and magnification in order to see it (most commonly found in first maxillary molars; studies have shown an average of 76% up to 96% of such teeth with the presence of an MB2 canal). This infected canal may cause a continued infection or "flare up" of the tooth. Any tooth may have more canals than expected, and these canals may be missed when the root canal is performed. Sometimes canals may be unusually shaped, making them impossible to clean and fill completely; some infected material may remain in the canal. Sometimes the canal filling does not fully extend to the apex of the tooth, or it does not fill the canal as densely as it should. Sometimes a tooth root may be perforated while the root canal is being treated, making it difficult to fill the tooth. The perforation may be filled with a root repair material, such as one derived from natural cement called MTA. A specialist can often re-treat failing root canals, and these teeth will then heal, often years after the initial root canal procedure.
However, the survival or functionality of the endodontically-treated tooth is often the most important aspect of endodontic treatment outcomes, rather than apical healing alone.[12] Recent studies indicate that substances commonly used to clean the root canal space incompletely sterilize the canal.[13] A properly restored tooth following root canal therapy yields long-term success rates near 97%. In a large scale Delta Dental Study of over 1.6 million patients who had root canal therapy, 97% had retained their teeth 8 years following the procedure, with most untoward events, such as re-treatment, apical surgery or extraction, occurring during the first 3 years after the initial endodontic treatment.[14] Endodontically treated teeth are prone to extraction mainly due to non-restorable carious destruction and to a lesser extent to endodontic-related reasons such as endodontic failure, vertical root fracture, or perforation (procedural error).[6]
Systemic issues An infected tooth may endanger other parts of the body. People with special vulnerabilities, such as prosthetic joint replacement or mitral valve prolapse, may need to take antibiotics to protect from infection spreading during dental procedures. Both endodontic therapy and tooth extraction can lead to subsequent jaw bone infection. The American Dental Association (ADA) asserts that any risks can be adequately controlled.
In the early 1900s, several researchers theorized that bacteria from teeth which had necrotic pulps or which had received endodontic treatment could cause chronic or local infection in areas distant from the tooth through the transfer of bacteria through the bloodstream. This was called the "focal infection theory", and it led some dentists to advocate dental extraction. In the 1930s, this theory was discredited, but the theory was recently revived by a book entitled Root Canal Cover-Up Exposed which used the early discredited research, and further complicated by epidemiological studies which found correlations between periodontal disease and heart disease, strokes, and preterm births. Bacteremia (bacteria in the bloodstream) can be caused by dental procedures, particularly after dental extractions, but endodontically treated teeth alone do not cause bacteremia or systemic disease.[
DENTAL IMPLANT
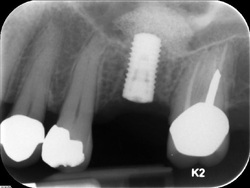
A dental implant is a "root" device, usually made of titanium, used in dentistry to support restorations that resemble a tooth or group of teeth to replace missing teeth.
Virtually all dental implants placed today are root-form endosseous implants, i.e., they appear similar to an actual tooth root (and thus possess a "root-form") and are placed within the bone (endo- being the Greek prefix for "in" and osseous referring to "bone"). The bone of the jaw accepts and osseointegrates with the titanium post. Osseointegration refers to the fusion of the implant surface with the surrounding bone. Dental implants will fuse with bone; however, they lack the periodontal ligament, so they will feel slightly different than natural teeth during chewing.
Prior to the advent of root-form endosseous implants, most implants were either blade endosseous implants, in that the shape of the metal piece placed within the bone resembled a flat blade, or subperiosteal implants, in which a framework was constructed to lie upon and was attached with screws to the exposed bone of the jaws.
Dental implants can be used to support a number of dental prostheses, including crowns, implant-supported bridges or dentures.[1] They can also be used as anchorage for orthodontic tooth movement. The use of dental implants permits undirectional tooth movement without reciprocal action.
Contents
In the 1950s research was being conducted at Cambridge University in England to study blood flow in vivo. These workers devised a method of constructing a chamber of titanium which was then embedded into the soft tissue of the ears of rabbits. In 1952 the Swedish orthopaedic surgeon, P I Brånemark, was interested in studying bone healing and regeneration, and adopted the Cambridge designed ‘rabbit ear chamber’ for use in the rabbit femur. Following several months of study he attempted to retrieve these expensive chambers from the rabbits and found that he was unable to remove them. Per Brånemark observed that bone had grown into such close proximity with the titanium that it effectively adhered to the metal. Brånemark carried out many further studies into this phenomenon, using both animal and human subjects, which all confirmed this unique property of titanium.
Dr. Leonard Linkow placed his first dental implant in 1952, four months after he graduated from dental school. By 1992, Dr. Linkow had placed over 19,000 dental implants and stopped counting. He retired from private practice in 2002 leaving a body of work that included 12 books and 36 patents. Many implant dentists refer to Dr. Linkow as the father of modern implant dentistry.[2]
Meanwhile an Italian medical doctor called Stefano Melchiade Tramonte, understood that titanium could be used for dental restorations and after designing a titanium screw to support his own dental prosthesis, started to use it on many patients in his clinic in 1959. The good results of his clinical studies on humans were published in 1966.[3]
Although Brånemark had originally considered that the first work should centre on knee and hip surgery, he finally decided that the mouth was more accessible for continued clinical observations and the high rate of edentulism in the general population offered more subjects for widespread study. He termed the clinically observed adherence of bone with titanium as ‘osseointegration’. In 1965 Brånemark, who was by then the Professor of Anatomy at Gothenburg University in Sweden, placed his first titanium dental implant into a human volunteer.
Contemporaneous independent research in the United States by Stevens and Alexander led to a 1969 US patent filing for titanium dental implants.[4]
Over the next fourteen years Brånemark published many studies on the use of titanium in dental implantology until in 1978 he entered into a commercial partnership with the Swedish defense company, Bofors AB for the development and marketing of his dental implants. With Bofors (later to become Nobel Industries) as the parent company, Nobelpharma AB (later to be renamed Nobel Biocare) was founded in 1981 to focus on dental implantology. To the present day over 7 million Brånemark System implants have now been placed and hundreds of other companies produce dental implants. The majority of dental implants currently available are shaped like small screws, with either tapered or parallel sides. They can be placed at the same time as a tooth is removed by engaging with the bone of the socket wall and sometimes also with the bone beyond the tip of the socket. Current evidence suggests that implants placed straight into an extraction socket have comparable success rates to those placed into healed bone.[5] The success rate and radiographic results of immediate restorations of dental implants placed in fresh extraction sockets (the temporary crowns placed at the same time) have been shown to be comparable to those obtained with delayed loading (the crowns placed weeks or months later) in carefully selected cases[6]
Some current research in dental implantology is focusing on the use of ceramic materials such as zirconia (ZrO2) in the manufacture of dental implants. Zirconia is the dioxide of zirconium, a metal close to titanium in the periodic table and with similar biocompatibility properties.[7] Although generally the same shape as titanium implants, zirconia, which has been used successfully for orthopaedic surgery for a number of years, has the advantage of being more cosmetically aesthetic owing to its bright tooth-like colour.[8] However, long-term clinical data is necessary before one-piece ZrO2 implants can be recommended for daily practice.[9]
Composition A typical implant consists of a titanium screw (resembling a tooth root) with a roughened or smooth surface. The majority of dental implants are made out of commercially pure titanium, which is available in 4 grades depending upon the amount of carbon and iron contained.[10] More recently grade 5 titanium has increased in use. Grade 5 titanium, Titanium 6AL-4V, (signifying the Titanium alloy containing 6% Aluminium and 4% Vanadium alloy) is believed to offer similar osseointegration levels as commercially pure titanium. Ti- 6Al-4V alloy offers better tensile strength and fracture resistance. Today most implants are still made out of commercially pure titanium (grades 1 to 4) but some implant systems are fabricated out of the Ti-6Al-4V alloy.[11] Implant surfaces may be modified by plasma spraying, anodizing,[12] etching, or sandblasting to increase the surface area and osseointegration potential of the implant. Recent studies have suggested that the chemical modification of the implant surface by polyelectrolyte coating [13] or nanoscale roughness manipulation [14] enhance cell proliferation and differentiation and promote osseointegration.
Training There is no specialty recognized by the ADA for dental implants in the United States. Implant surgery may be performed as an outpatient under general anesthesia, oral conscious sedation, nitrous oxide sedation, intravenous sedation or under local anesthesia by trained and certified clinicians including general dentists, endodontists, oral surgeons, periodontists, and prosthodontists.
The legal training requirements for dentists who carry out implant treatment differ from country to country. In the UK, implant dentistry is considered by the General Dental Council to be a postgraduate sphere of dentistry. In other words it is not sufficiently covered during the teaching of the university dental degree course and dentists wishing to practice in dental implantology legally need to undergo additional formal postgraduate training. The General Dental Council has published strict guidelines on the training required for a dentist to be able to place dental implants in general dental practice.[15] UK dentists need to complete a competency assessed postgraduate extended learning program before providing implant dentistry to patients.
The degree to which both graduate and post-graduate dentists receive training in the surgical placement of implants varies from country to country,[16][17][18] but it seems likely that lack of formal training will lead to higher complication rates.[19]
In the United States, several implant training courses are available through most major manufacturers. These hands-on courses emphasize treatment planning, case selection, implant placement protocol, restorative techniques, and marketing strategies.
Surgical procedure Surgical planning Prior to commencement of surgery, careful and detailed planning is required to identify vital structures such as the inferior alveolar nerve or the sinus, as well as the shape and dimensions of the bone to properly orient the implants for the most predictable outcome. Two-dimensional radiographs, such as orthopantomographs or periapicals are often taken prior to the surgery. Sometimes, a CT scan will also be obtained. Specialized 3D CAD/CAM computer programs may be used to plan the case.
Whether CT-guided or manual, a 'stent' may sometimes be used to facilitate the placement of implants. A surgical stent is an acrylic wafer that fits over either the teeth, the bone surface or the mucosa (when all the teeth are missing) with pre-drilled holes to show the position and angle of the implants to be placed. The surgical stent may be produced using stereolithography following computerized planning of a case from the CT scan. CT guided surgery may double the cost compared to more commonly accepted approaches.
Basic procedure In its most basic form, the placement of an implant requires a preparation into the bone using either hand osteotomes or precision drills with highly regulated speed[20] to prevent burning or pressure necrosis of the bone. After a variable amount of time to allow the bone to grow on to the surface of the implant (osseointegration), a crown or crowns can be placed on the implant. Unlike conventional dental implants, Mini dental implants may be loaded immediately and still have a high survival rate (94%).[21][22] The amount of time required to place an implant will vary depending on the experience of the practitioner, the quality and quantity of the bone and the difficulty of the individual situation.
Detail procedure At edentulous (without teeth) jaw sites, a pilot hole is bored into the recipient bone, taking care to avoid the vital structures (in particular the inferior alveolar nerve or IAN and the mental foramen within the mandible). Drilling into jawbone usually occurs in several separate steps. The pilot hole is expanded by using progressively wider drills (typically between three and seven successive drilling steps, depending on implant width and length). Care is taken not to damage the osteoblast or bone cells by overheating. A cooling saline or water spray keeps the temperature of the bone to below 47 degrees Celsius (approximately 117 degrees Fahrenheit). The implant screw can be self-tapping, and is screwed into place at a precise torque so as not to overload the surrounding bone (overloaded bone can die, a condition called osteonecrosis, which may lead to failure of the implant to fully integrate or bond with the jawbone). Typically in most implant systems, the osteotomy or drilled hole is about 1mm deeper than the implant being placed, due to the shape of the drill tip. Surgeons must take the added length into consideration when drilling in the vicinity of vital structures.
Surgical incisions Straumann guide pin in place to visualize positioning, angulation and depth prior to implant placement. Implant is being placed as a flapless procedure because it was clinically and radiographically evident that there is adequate bone in all dimensions. Traditionally, an incision is made over the crest of the site where the implant is to be placed. This is referred to as a 'flap'. Some systems allow for 'flapless' surgery where a piece of mucosa is punched-out from over the implant site. Proponents of 'flapless' surgery believe that it decreases recovery time while its detractors believe it increases complication rates because the edge of bone cannot be visualized.[23][24] Because of these visualization problems flapless surgery is often carried out using a surgical guide constructed following computerized 3D planning of a pre-operative CT scan.
Healing time The amount of time required for an implant to become osseointegrated is a hotly debated topic.[25] Consequently the amount of time that practitioners allow the implant to heal before placing a restoration on it varies widely. In general, practitioners allow 2–6 months for healing but preliminary studies show that early loading of implant may not increase early or long term complications.[26] Minimally invasive methods of early dental implant placement reduce the cost of installed implants and shortens the implant-prosthetic rehabilitation time with 4–6 months.[27] If the implant is loaded too soon, it is possible that the implant may move which results in failure. For conventional implants, the subsequent time to heal, possibly graft, and eventually place a new implant may take up to eighteen months. For this reason many are reluctant to push the envelope for healing.
One-stage, two-stage surgery When an implant is placed either a 'healing abutment', which comes through the mucosa, is placed or a 'cover screw' which is flush with the surface of the dental implant is placed. When a cover screw is placed the mucosa covers the implant while it integrates then a second surgery is completed to place the healing abutment.
Two-stage surgery is sometimes chosen when a concurrent bone graft is placed or surgery on the mucosa may be required for esthetic reasons. The latter is usually important where an implant is placed in the "esthetic zone". This allows more control over the healing and as a result the predictability of the final result.[28] Some implants are one piece so that no healing abutment is required.
In carefully selected cases, patients can be implanted and restored in a single surgery, in a procedure labeled "Immediate Loading". In such cases a provisional prosthetic tooth or crown is shaped to avoid the force of the bite transferring to the implant while it integrates with the bone.
Surgical timing There are different approaches to place dental implants after tooth extraction. The approaches are:
Most data suggests that when placed into single rooted tooth sites with healthy bone and mucosa around them, the success rates are comparable to that of delayed procedures with no additional complications.[30]
Teeth in a Day "Teeth in a day", "All-on-four",[31] "Fast and fixed" are similar surgical concepts whereby implants are placed on the same day and a fixed prosthesis is attached to them. This allows the patient to leave with a fixed solution as opposed having to make do with a removal temporary prosthesis whilst the implant osseointegrate with the bone. This concept is appropriate for completely edentulous jaws where either the teeth are to be extracted or have already been removed.
Use of CT scanning CT scan of the lower jaw. This shows eight dental implants superimposed over the lower jaw in areas of maximum bone and four teeth that will be extracted.[32] When computed tomography or, more specifically, cone beam computed tomography or CBCT (3D X-ray imaging) is used preoperatively to accurately pinpoint vital structures including the inferior alveolar canal, the mental foramen, and the maxillary sinus, the chances of complications might be reduced as is chairtime and number of visits.[33] Cone beam CT scanning, when compared to traditional medical CT scanning, utilizes less than 2% of the radiation, provides more accuracy in the area of interest, and is safer for the patient.[34] CBCT allows the surgeon to create a surgical guide, which allows the surgeon to accurately angle the implant into the ideal space.[35]
Complementary procedures Sinus lifting is a common surgical intervention. A dentist or specialist with proper training such as an oral surgeon, periodontist, general dentist or prosthodontist, thickens the inadequate part of atrophic maxilla towards the sinus with the help of bone transplantation or bone expletive substance. This results in more volume for a better quality bone site for the implantation. Prudent clinicians who wish to avoid placement of implants into the sinus cavity pre-plan sinus lift surgery using the CBCT X-ray, as in the case of posterior mandibular implants discussed earlier. Sinus lift is done to reestablish bone to your upper jaw, in the missing tooth area of molars and premolars. This allows for dental implants to be placed in the bone of the upper jaw.[36]
Bone grafting will be necessary in cases where there is a lack of adequate maxillary or mandibular bone in terms of front to back (lip to tongue) depth or thickness; top to bottom height; and left to right width. Sufficient bone is needed in three dimensions to securely integrate with the root-like implant. Improved bone height—which is very difficult to achieve—is particularly important to assure ample anchorage of the implant's root-like shape because it has to support the mechanical stress of chewing, just like a natural tooth.
Typically, implantologists try to place implants at least as deeply into bone as the crown or tooth will be above the bone. This is called a 1:1 crown to root ratio. This ratio establishes the target for bone grafting in most cases. If 1:1 or more cannot be achieved, the patient is usually advised that only a short implant can be placed and to not expect a long period of usability.
A wide range of grafting materials and substances may be used during the process of bone grafting / bone replacement. They include the patient's own bone (autograft), which may be harvested from the hip (iliac crest) or from spare jawbone; processed bone from cadavers (allograft)Which is demineralised; bovine bone or coral (xenograft); or artificially produced bone-like substances (calcium sulfate with names like Regeneform; and hydroxyapatite or HA, which is the primary form of calcium found in bone) or calcium phosphosilicate which is available in a mouldable putty form. The HA is effective as a substrate for osteoblasts to grow on. Some implants are coated with HA for this reason, although the bone forming properties of many of these substances is a hotly debated topic in bone research groups. Alternatively the bone intended to support the implant can be split and widened with the implant placed between the two halves like a sandwich. This is referred to as a 'ridge split' procedure.
Bone graft surgery has its own standard of care. In a typical procedure, the clinician creates a large flap of the gingiva or gum to fully expose the jawbone at the graft site, performs one or several types of block and onlay grafts in and on existing bone, then installs a membrane designed to repel unwanted infection-causing microbiota found in the oral cavity. Then the mucosa is carefully sutured over the site. Together with a course of systemic antibiotics and topical antibacterial mouth rinses, the graft site is allowed to heal (several months).
The clinician typically takes a new radiograph to confirm graft success in width and height, and assumes that positive signs in these two dimensions safely predict success in the third dimension; depth. Where more precision is needed, usually when mandibular implants are being planned, a 3D or cone beam radiograph may be called for at this point to enable accurate measurement of bone and location of nerves and vital structures for proper treatment planning. The same radiographic data set can be employed for the preparation of computer-designed placement guides.
Correctly performed, a bone graft produces live vascular bone which is very much like natural jawbone and is therefore suitable as a foundation for implants.
Considerations Chrome-cobalt disc with bridges and crowns for dental implants manufactured using WorkNC Dental CAD/CAM For dental implant procedure to work, there must be enough bone in the jaw, and the bone has to be strong enough to hold and support the implant. If there is not enough bone, more may need to be added with a bone graft procedure discussed earlier. Sometimes, this procedure is called bone augmentation, or guided bone regeneration. Mini dental implants are particularly useful in the endentulous arch with minimal remaining bone facio-lingually.[37] In addition, natural teeth and supporting tissues near where the implant will be placed must be in good health.
In all cases careful consideration must be given to the final functional aspects of the restoration, such as assessing the forces which will be placed on the implant. Implant loading from chewing and parafunction (abnormal grinding or clenching habits) can exceed the biomechanic tolerance of the implant bone interface and/or the titanium material itself, causing failure. This can be failure of the implant itself (fracture) or bone loss, a "melting" or resorption of the surrounding bone.
The dentist must first determine what type of prosthesis will be fabricated. Only then can the specific implant requirements including number, length, diameter, and thread pattern be determined. In other words, the case must be reverse engineered by the restoring dentist prior to the surgery. If bone volume or density is inadequate, a bone graft procedure must be considered first. The restoring dentist may consult with the oral surgeon, periodontist, endodontist, or another trained general dentist to co-treat the patient. Usually, physical models or impressions of the patient's jawbones and teeth are made by the restorative dentist at the implant surgeons request, and are used as physical aids to treatment planning. If not supplied, the implant surgeon makes his own or relies upon advanced computer-assisted tomography or a cone beam CT scan to achieve the proper treatment plan.
Computer simulation software based on CT scan data allows virtual implant surgical placement based on a barium impregnated prototype of the final prosthesis. This predicts vital anatomy, bone quality, implant characteristics, the need for bone grafting, and maximizing the implant bone surface area for the treatment case creating a high level of predictability. Computer CAD/CAM milled or stereolithography based drill guides can be developed for the implant surgeon to facilitate proper implant placement based on the final prosthesis' occlusion and aesthetics.
Treatment planning software can also be used to demonstrate "try-ins" to the patient on a computer screen. When options have been fully discussed between patient and surgeon, the same software can be used to produce precision drill guides. Specialized software applications such as 'SimPlant' (simulated implant) or 'NobelGuide' use the digital data from a patient's CBCT to build a treatment plan. A data set is then produced and sent to a lab for production of a precision in-mouth drilling guide.[38]
Success rates Dental implant success is related to operator skill, quality and quantity of the bone available at the site, and the patient's oral hygiene. The consensus is that implants carry a success rate of around 75%[39]
One of the most important factors that determine implant success is the achievement and maintenance of implant stability.[40] The stability is presented as an ISQ (Implant Stability Quotient) value. Other contributing factors to the success of dental implant placement, as with most surgical procedures, include the patient's overall general health and compliance with post-surgical care.
Dental implants can help stop periodontal diseases from forming. This is because gaps formed by missing teeth can cause periodontal disease and infections that may affect a person’s ability to eat or speak. It is very important to fill in any gaps to prevent the adjacent and opposing set of teeth from shifting positions or causing further damage to the mouth.[41]
Failure Failure of a dental implant is often related to the failure of the implant to osseointegrate correctly with the bone, or vice-versa. A dental implant is considered to be a failure if it is lost, mobile or shows peri-implant (around the implant) bone loss of greater than 1.0 mm in the first year and greater than 0.2 mm a year after.
Dental implants are not susceptible to dental caries but they can develop a condition called peri-implantitis. This is an inflammatory condition of the mucosa and/or bone around the implant which may result in bone loss and eventual loss of the implant. The condition is usually, but not always, associated with a chronic infection. Peri-implantitis is more likely to occur in heavy smokers, patients with diabetes, patients with poor oral hygiene and cases where the mucosa around the implant is thin.[42]
Currently there is no universal agreement on the best treatment for peri-implantitis. The condition and its causes is still poorly understood.[43]
Risk of failure is increased in smokers. For this reason implants are frequently placed only after a patient has stopped smoking as the treatment is very expensive. More rarely, an implant may fail because of poor positioning at the time of surgery, or may be overloaded initially causing failure to integrate. If smoking and positioning problems exist prior to implant surgery, clinicians often advise patients that a bridge or partial denture rather than an implant may be a better solution.
Failure may also occur independently of the causes outlined above. Implants like any other object suffers from wear and tear. If the implant(s)in question are replacing commonly used teeth, then these may suffer from wear and tear and after years may crack and break up, although this is a very rare occurrence.
In the majority of cases where an implant fails to integrate with the bone and is rejected by the body the cause is unknown. This may occur in around 5% of cases. To this day we still do not know why bone will integrate with titanium dental implants and why it does not reject the material as a 'foreign body'. Many theories have been postulated over the last five decades. A recent theory argues that rather than being an active biological tissue response, the integration of bone with an implant is the lack of a negative tissue response. In other words, for unknown reasons the usual response of the body to reject foreign objects implanted into it does not function correctly with titanium implants. It has further been postulated that an implant rejection occurs in patients whose bone tissues actually react as they naturally should with the 'foreign body' and reject the implant in the same manner that would occur with most other implanted materials.[44]
Contraindications There are few absolute contraindications to implant dentistry. However, there are some systemic, behavioral, and anatomic considerations that should be assessed.
Particularly for mandibular (lower jaw) implants, in the vicinity of the mental foramen (MF), there must be sufficient alveolar bone above the mandibular canal also called the inferior alveolar canal or IAC (which acts as the conduit for the neurovascular bundle carrying the inferior alveolar nerve or IAN).
Failure to precisely locate the IAN and MF invites surgical insult by the drills and the implant itself. Such insult may cause irreparable damage to the nerve, often felt as a paresthesia (numbness) or dysesthesia (painful numbness) of the gum, lip and chin. This condition may persist for life and may be accompanied by unconscious drooling.
Uncontrolled Type II diabetes is a significant relative contraindication as healing following any type of surgical procedure is delayed due to poor peripheral blood circulation. Anatomic considerations include the volume and height of bone available. Often an ancillary procedure known as a block graft or sinus augmentation are needed to provide enough bone for successful implant placement.
There is new information about intravenous and oral bisphosphonates (taken for certain forms of breast cancer and osteoporosis, respectively) which may put patients at a higher risk of developing a delayed healing syndrome called osteonecrosis. Implants are contraindicated for some patients who take intravenous bisphosphonates.
The many millions of patients who take an oral bisphosphonate (such as Actonel, Fosamax and Boniva) may sometimes be advised to stop the administration prior to implant surgery, then resume several months later. However, current evidence suggests that this protocol may not be necessary. As of January 2008, an oral bisphosphonate study reported in the February 2008 Journal of Oral and Maxillofacial Surgery, reviewing 115 cases that included 468 implants, concluded "There is no evidence of bisphosphonate-associated osteonecrosis of the jaw in any of the patients evaluated in the clinic and those contacted by phone or e-mail reported no symptoms."[45]
The American Dental Association had addressed bisphosphonates in an article entitled "Bisphosphonate medications and your oral health,"[46] In an Overview, the ADA stated "The risk of developing BON [bisphosphonate-associated osteonecrosis of the jaw] in patients on oral bisphosphonate therapy appears to be very low...". The ADA Council on Scientific Affairs also employed a panel of experts who issued recommendations [for clinicians] for treatment of patients on oral bisphosphonates, published in June 2006. The overview may be read online at ada.org but it has now been superseded by a huge study—encompassing over 700,000 cases—entitled "Bisphosphonate Use and the Risk of Adverse Jaw Outcomes." Like the 2008 JOMS study, the ADA study exonerates oral bisphosphonates as a contraindication to dental implants.[47]
Bruxism (tooth clenching or grinding) is another consideration which may reduce the prognosis for treatment. The forces generated during bruxism are particularly detrimental to implants while bone is healing; micromovements in the implant positioning are associated with increased rates of implant failure. Bruxism continues to pose a threat to implants throughout the life of the recipient.[48] Natural teeth contain a periodontal ligament allowing each tooth to move and absorb shock in response to vertical and horizontal forces. Once replaced by dental implants, this ligament is lost and teeth are immovably anchored directly into the jaw bone. This problem can be minimized by wearing a custom made mouthguard (such an NTI appliance) at night.
Postoperatively, after implants have been placed, there are physical contraindications that prompt rapid action by the implantology team. Excessive or severe pain lasting more than three days is a warning sign, as is excessive bleeding. Constant numbness of the gingiva (gum), lip and chin—usually noticed after surgical anesthesia wears off—is another warning sign. In the latter case, which may be accompanied by severe constant pain, the standard of care calls for diagnosis to determine if the surgical procedure insulted the IAN. A 3D cone beam X-ray provides the necessary data, but even before this step a prudent implantologist may back out or completely remove an implant in an effort to restore nerve function because delay is usually ineffective. Depending upon the evidence visible with a 3D X-ray, patients may be referred to a specialist in nerve repair. In all cases, speed in diagnosis and treatment are necessary.
Market In the United States and the United Kingdom, there is no exclusive specialty in 'implantology'.
Any practitioner who carries out implant treatment, whether in the surgical insertion or the final provision of the prosthesis, must be adequately trained. Legal training requirements differ between countries.
In 2008, in the UK the General Dental Council (GDC) laid down strict training requirements[49] for dentists involved in dental implantology. Any dentist in the UK who wishes to train in the field of dental implantology must take part in an extended learning program which covers a detailed theory syllabus, as approved by the GDC,[50] in addition to formal supervised surgical training and mentoring. Dentists must not take part in implant dentistry in the UK until they have been approved by the training provider as having passed a formal competency assessment. Failure to comply with the GDC regulations may result in a dentist being removed from the Dental Register and hence losing the right to practice dentistry in the UK.[51]
The fast growing demand for prosthetic solutions based upon dental implants caused the birth of hundreds of different brands available on the market, about 150 in the UK[52] and more than 300 in Italy,[53] but only a very few of these have enough documentation and clinical practice for being recognized worldwide.[53] According to a study published in the Clinical Implant Dentistry and Related Research, only three brands of implants were tested by at least two independent (not sponsored) clinical researches,[54] which should push the patients to ask for more information before accepting any treatment, and to demand for a real implant passport.
Virtually all dental implants placed today are root-form endosseous implants, i.e., they appear similar to an actual tooth root (and thus possess a "root-form") and are placed within the bone (endo- being the Greek prefix for "in" and osseous referring to "bone"). The bone of the jaw accepts and osseointegrates with the titanium post. Osseointegration refers to the fusion of the implant surface with the surrounding bone. Dental implants will fuse with bone; however, they lack the periodontal ligament, so they will feel slightly different than natural teeth during chewing.
Prior to the advent of root-form endosseous implants, most implants were either blade endosseous implants, in that the shape of the metal piece placed within the bone resembled a flat blade, or subperiosteal implants, in which a framework was constructed to lie upon and was attached with screws to the exposed bone of the jaws.
Dental implants can be used to support a number of dental prostheses, including crowns, implant-supported bridges or dentures.[1] They can also be used as anchorage for orthodontic tooth movement. The use of dental implants permits undirectional tooth movement without reciprocal action.
Contents
- 1 History
- 2 Composition
- 3 Training
- 4 Surgical procedure
- 5 Complementary procedures
- 6 Considerations
- 7 Success rates
- 8 Failure
- 9 Contraindications
- 10 Market
- 11 Prices
- 12 See also
- 13 References
- 14 External links
In the 1950s research was being conducted at Cambridge University in England to study blood flow in vivo. These workers devised a method of constructing a chamber of titanium which was then embedded into the soft tissue of the ears of rabbits. In 1952 the Swedish orthopaedic surgeon, P I Brånemark, was interested in studying bone healing and regeneration, and adopted the Cambridge designed ‘rabbit ear chamber’ for use in the rabbit femur. Following several months of study he attempted to retrieve these expensive chambers from the rabbits and found that he was unable to remove them. Per Brånemark observed that bone had grown into such close proximity with the titanium that it effectively adhered to the metal. Brånemark carried out many further studies into this phenomenon, using both animal and human subjects, which all confirmed this unique property of titanium.
Dr. Leonard Linkow placed his first dental implant in 1952, four months after he graduated from dental school. By 1992, Dr. Linkow had placed over 19,000 dental implants and stopped counting. He retired from private practice in 2002 leaving a body of work that included 12 books and 36 patents. Many implant dentists refer to Dr. Linkow as the father of modern implant dentistry.[2]
Meanwhile an Italian medical doctor called Stefano Melchiade Tramonte, understood that titanium could be used for dental restorations and after designing a titanium screw to support his own dental prosthesis, started to use it on many patients in his clinic in 1959. The good results of his clinical studies on humans were published in 1966.[3]
Although Brånemark had originally considered that the first work should centre on knee and hip surgery, he finally decided that the mouth was more accessible for continued clinical observations and the high rate of edentulism in the general population offered more subjects for widespread study. He termed the clinically observed adherence of bone with titanium as ‘osseointegration’. In 1965 Brånemark, who was by then the Professor of Anatomy at Gothenburg University in Sweden, placed his first titanium dental implant into a human volunteer.
Contemporaneous independent research in the United States by Stevens and Alexander led to a 1969 US patent filing for titanium dental implants.[4]
Over the next fourteen years Brånemark published many studies on the use of titanium in dental implantology until in 1978 he entered into a commercial partnership with the Swedish defense company, Bofors AB for the development and marketing of his dental implants. With Bofors (later to become Nobel Industries) as the parent company, Nobelpharma AB (later to be renamed Nobel Biocare) was founded in 1981 to focus on dental implantology. To the present day over 7 million Brånemark System implants have now been placed and hundreds of other companies produce dental implants. The majority of dental implants currently available are shaped like small screws, with either tapered or parallel sides. They can be placed at the same time as a tooth is removed by engaging with the bone of the socket wall and sometimes also with the bone beyond the tip of the socket. Current evidence suggests that implants placed straight into an extraction socket have comparable success rates to those placed into healed bone.[5] The success rate and radiographic results of immediate restorations of dental implants placed in fresh extraction sockets (the temporary crowns placed at the same time) have been shown to be comparable to those obtained with delayed loading (the crowns placed weeks or months later) in carefully selected cases[6]
Some current research in dental implantology is focusing on the use of ceramic materials such as zirconia (ZrO2) in the manufacture of dental implants. Zirconia is the dioxide of zirconium, a metal close to titanium in the periodic table and with similar biocompatibility properties.[7] Although generally the same shape as titanium implants, zirconia, which has been used successfully for orthopaedic surgery for a number of years, has the advantage of being more cosmetically aesthetic owing to its bright tooth-like colour.[8] However, long-term clinical data is necessary before one-piece ZrO2 implants can be recommended for daily practice.[9]
Composition A typical implant consists of a titanium screw (resembling a tooth root) with a roughened or smooth surface. The majority of dental implants are made out of commercially pure titanium, which is available in 4 grades depending upon the amount of carbon and iron contained.[10] More recently grade 5 titanium has increased in use. Grade 5 titanium, Titanium 6AL-4V, (signifying the Titanium alloy containing 6% Aluminium and 4% Vanadium alloy) is believed to offer similar osseointegration levels as commercially pure titanium. Ti- 6Al-4V alloy offers better tensile strength and fracture resistance. Today most implants are still made out of commercially pure titanium (grades 1 to 4) but some implant systems are fabricated out of the Ti-6Al-4V alloy.[11] Implant surfaces may be modified by plasma spraying, anodizing,[12] etching, or sandblasting to increase the surface area and osseointegration potential of the implant. Recent studies have suggested that the chemical modification of the implant surface by polyelectrolyte coating [13] or nanoscale roughness manipulation [14] enhance cell proliferation and differentiation and promote osseointegration.
Training There is no specialty recognized by the ADA for dental implants in the United States. Implant surgery may be performed as an outpatient under general anesthesia, oral conscious sedation, nitrous oxide sedation, intravenous sedation or under local anesthesia by trained and certified clinicians including general dentists, endodontists, oral surgeons, periodontists, and prosthodontists.
The legal training requirements for dentists who carry out implant treatment differ from country to country. In the UK, implant dentistry is considered by the General Dental Council to be a postgraduate sphere of dentistry. In other words it is not sufficiently covered during the teaching of the university dental degree course and dentists wishing to practice in dental implantology legally need to undergo additional formal postgraduate training. The General Dental Council has published strict guidelines on the training required for a dentist to be able to place dental implants in general dental practice.[15] UK dentists need to complete a competency assessed postgraduate extended learning program before providing implant dentistry to patients.
The degree to which both graduate and post-graduate dentists receive training in the surgical placement of implants varies from country to country,[16][17][18] but it seems likely that lack of formal training will lead to higher complication rates.[19]
In the United States, several implant training courses are available through most major manufacturers. These hands-on courses emphasize treatment planning, case selection, implant placement protocol, restorative techniques, and marketing strategies.
Surgical procedure Surgical planning Prior to commencement of surgery, careful and detailed planning is required to identify vital structures such as the inferior alveolar nerve or the sinus, as well as the shape and dimensions of the bone to properly orient the implants for the most predictable outcome. Two-dimensional radiographs, such as orthopantomographs or periapicals are often taken prior to the surgery. Sometimes, a CT scan will also be obtained. Specialized 3D CAD/CAM computer programs may be used to plan the case.
Whether CT-guided or manual, a 'stent' may sometimes be used to facilitate the placement of implants. A surgical stent is an acrylic wafer that fits over either the teeth, the bone surface or the mucosa (when all the teeth are missing) with pre-drilled holes to show the position and angle of the implants to be placed. The surgical stent may be produced using stereolithography following computerized planning of a case from the CT scan. CT guided surgery may double the cost compared to more commonly accepted approaches.
Basic procedure In its most basic form, the placement of an implant requires a preparation into the bone using either hand osteotomes or precision drills with highly regulated speed[20] to prevent burning or pressure necrosis of the bone. After a variable amount of time to allow the bone to grow on to the surface of the implant (osseointegration), a crown or crowns can be placed on the implant. Unlike conventional dental implants, Mini dental implants may be loaded immediately and still have a high survival rate (94%).[21][22] The amount of time required to place an implant will vary depending on the experience of the practitioner, the quality and quantity of the bone and the difficulty of the individual situation.
Detail procedure At edentulous (without teeth) jaw sites, a pilot hole is bored into the recipient bone, taking care to avoid the vital structures (in particular the inferior alveolar nerve or IAN and the mental foramen within the mandible). Drilling into jawbone usually occurs in several separate steps. The pilot hole is expanded by using progressively wider drills (typically between three and seven successive drilling steps, depending on implant width and length). Care is taken not to damage the osteoblast or bone cells by overheating. A cooling saline or water spray keeps the temperature of the bone to below 47 degrees Celsius (approximately 117 degrees Fahrenheit). The implant screw can be self-tapping, and is screwed into place at a precise torque so as not to overload the surrounding bone (overloaded bone can die, a condition called osteonecrosis, which may lead to failure of the implant to fully integrate or bond with the jawbone). Typically in most implant systems, the osteotomy or drilled hole is about 1mm deeper than the implant being placed, due to the shape of the drill tip. Surgeons must take the added length into consideration when drilling in the vicinity of vital structures.
Surgical incisions Straumann guide pin in place to visualize positioning, angulation and depth prior to implant placement. Implant is being placed as a flapless procedure because it was clinically and radiographically evident that there is adequate bone in all dimensions. Traditionally, an incision is made over the crest of the site where the implant is to be placed. This is referred to as a 'flap'. Some systems allow for 'flapless' surgery where a piece of mucosa is punched-out from over the implant site. Proponents of 'flapless' surgery believe that it decreases recovery time while its detractors believe it increases complication rates because the edge of bone cannot be visualized.[23][24] Because of these visualization problems flapless surgery is often carried out using a surgical guide constructed following computerized 3D planning of a pre-operative CT scan.
Healing time The amount of time required for an implant to become osseointegrated is a hotly debated topic.[25] Consequently the amount of time that practitioners allow the implant to heal before placing a restoration on it varies widely. In general, practitioners allow 2–6 months for healing but preliminary studies show that early loading of implant may not increase early or long term complications.[26] Minimally invasive methods of early dental implant placement reduce the cost of installed implants and shortens the implant-prosthetic rehabilitation time with 4–6 months.[27] If the implant is loaded too soon, it is possible that the implant may move which results in failure. For conventional implants, the subsequent time to heal, possibly graft, and eventually place a new implant may take up to eighteen months. For this reason many are reluctant to push the envelope for healing.
One-stage, two-stage surgery When an implant is placed either a 'healing abutment', which comes through the mucosa, is placed or a 'cover screw' which is flush with the surface of the dental implant is placed. When a cover screw is placed the mucosa covers the implant while it integrates then a second surgery is completed to place the healing abutment.
Two-stage surgery is sometimes chosen when a concurrent bone graft is placed or surgery on the mucosa may be required for esthetic reasons. The latter is usually important where an implant is placed in the "esthetic zone". This allows more control over the healing and as a result the predictability of the final result.[28] Some implants are one piece so that no healing abutment is required.
In carefully selected cases, patients can be implanted and restored in a single surgery, in a procedure labeled "Immediate Loading". In such cases a provisional prosthetic tooth or crown is shaped to avoid the force of the bite transferring to the implant while it integrates with the bone.
Surgical timing There are different approaches to place dental implants after tooth extraction. The approaches are:
- Immediate post-extraction implant placement.
- Delayed immediate post-extraction implant placement (2 weeks to 3 months after extraction).
- Late implantation (3 months or more after tooth extraction).
- Immediate loading procedure.
- Early loading (1 week to 12 weeks).
- Delayed loading (over 3 months)
Most data suggests that when placed into single rooted tooth sites with healthy bone and mucosa around them, the success rates are comparable to that of delayed procedures with no additional complications.[30]
Teeth in a Day "Teeth in a day", "All-on-four",[31] "Fast and fixed" are similar surgical concepts whereby implants are placed on the same day and a fixed prosthesis is attached to them. This allows the patient to leave with a fixed solution as opposed having to make do with a removal temporary prosthesis whilst the implant osseointegrate with the bone. This concept is appropriate for completely edentulous jaws where either the teeth are to be extracted or have already been removed.
Use of CT scanning CT scan of the lower jaw. This shows eight dental implants superimposed over the lower jaw in areas of maximum bone and four teeth that will be extracted.[32] When computed tomography or, more specifically, cone beam computed tomography or CBCT (3D X-ray imaging) is used preoperatively to accurately pinpoint vital structures including the inferior alveolar canal, the mental foramen, and the maxillary sinus, the chances of complications might be reduced as is chairtime and number of visits.[33] Cone beam CT scanning, when compared to traditional medical CT scanning, utilizes less than 2% of the radiation, provides more accuracy in the area of interest, and is safer for the patient.[34] CBCT allows the surgeon to create a surgical guide, which allows the surgeon to accurately angle the implant into the ideal space.[35]
Complementary procedures Sinus lifting is a common surgical intervention. A dentist or specialist with proper training such as an oral surgeon, periodontist, general dentist or prosthodontist, thickens the inadequate part of atrophic maxilla towards the sinus with the help of bone transplantation or bone expletive substance. This results in more volume for a better quality bone site for the implantation. Prudent clinicians who wish to avoid placement of implants into the sinus cavity pre-plan sinus lift surgery using the CBCT X-ray, as in the case of posterior mandibular implants discussed earlier. Sinus lift is done to reestablish bone to your upper jaw, in the missing tooth area of molars and premolars. This allows for dental implants to be placed in the bone of the upper jaw.[36]
Bone grafting will be necessary in cases where there is a lack of adequate maxillary or mandibular bone in terms of front to back (lip to tongue) depth or thickness; top to bottom height; and left to right width. Sufficient bone is needed in three dimensions to securely integrate with the root-like implant. Improved bone height—which is very difficult to achieve—is particularly important to assure ample anchorage of the implant's root-like shape because it has to support the mechanical stress of chewing, just like a natural tooth.
Typically, implantologists try to place implants at least as deeply into bone as the crown or tooth will be above the bone. This is called a 1:1 crown to root ratio. This ratio establishes the target for bone grafting in most cases. If 1:1 or more cannot be achieved, the patient is usually advised that only a short implant can be placed and to not expect a long period of usability.
A wide range of grafting materials and substances may be used during the process of bone grafting / bone replacement. They include the patient's own bone (autograft), which may be harvested from the hip (iliac crest) or from spare jawbone; processed bone from cadavers (allograft)Which is demineralised; bovine bone or coral (xenograft); or artificially produced bone-like substances (calcium sulfate with names like Regeneform; and hydroxyapatite or HA, which is the primary form of calcium found in bone) or calcium phosphosilicate which is available in a mouldable putty form. The HA is effective as a substrate for osteoblasts to grow on. Some implants are coated with HA for this reason, although the bone forming properties of many of these substances is a hotly debated topic in bone research groups. Alternatively the bone intended to support the implant can be split and widened with the implant placed between the two halves like a sandwich. This is referred to as a 'ridge split' procedure.
Bone graft surgery has its own standard of care. In a typical procedure, the clinician creates a large flap of the gingiva or gum to fully expose the jawbone at the graft site, performs one or several types of block and onlay grafts in and on existing bone, then installs a membrane designed to repel unwanted infection-causing microbiota found in the oral cavity. Then the mucosa is carefully sutured over the site. Together with a course of systemic antibiotics and topical antibacterial mouth rinses, the graft site is allowed to heal (several months).
The clinician typically takes a new radiograph to confirm graft success in width and height, and assumes that positive signs in these two dimensions safely predict success in the third dimension; depth. Where more precision is needed, usually when mandibular implants are being planned, a 3D or cone beam radiograph may be called for at this point to enable accurate measurement of bone and location of nerves and vital structures for proper treatment planning. The same radiographic data set can be employed for the preparation of computer-designed placement guides.
Correctly performed, a bone graft produces live vascular bone which is very much like natural jawbone and is therefore suitable as a foundation for implants.
Considerations Chrome-cobalt disc with bridges and crowns for dental implants manufactured using WorkNC Dental CAD/CAM For dental implant procedure to work, there must be enough bone in the jaw, and the bone has to be strong enough to hold and support the implant. If there is not enough bone, more may need to be added with a bone graft procedure discussed earlier. Sometimes, this procedure is called bone augmentation, or guided bone regeneration. Mini dental implants are particularly useful in the endentulous arch with minimal remaining bone facio-lingually.[37] In addition, natural teeth and supporting tissues near where the implant will be placed must be in good health.
In all cases careful consideration must be given to the final functional aspects of the restoration, such as assessing the forces which will be placed on the implant. Implant loading from chewing and parafunction (abnormal grinding or clenching habits) can exceed the biomechanic tolerance of the implant bone interface and/or the titanium material itself, causing failure. This can be failure of the implant itself (fracture) or bone loss, a "melting" or resorption of the surrounding bone.
The dentist must first determine what type of prosthesis will be fabricated. Only then can the specific implant requirements including number, length, diameter, and thread pattern be determined. In other words, the case must be reverse engineered by the restoring dentist prior to the surgery. If bone volume or density is inadequate, a bone graft procedure must be considered first. The restoring dentist may consult with the oral surgeon, periodontist, endodontist, or another trained general dentist to co-treat the patient. Usually, physical models or impressions of the patient's jawbones and teeth are made by the restorative dentist at the implant surgeons request, and are used as physical aids to treatment planning. If not supplied, the implant surgeon makes his own or relies upon advanced computer-assisted tomography or a cone beam CT scan to achieve the proper treatment plan.
Computer simulation software based on CT scan data allows virtual implant surgical placement based on a barium impregnated prototype of the final prosthesis. This predicts vital anatomy, bone quality, implant characteristics, the need for bone grafting, and maximizing the implant bone surface area for the treatment case creating a high level of predictability. Computer CAD/CAM milled or stereolithography based drill guides can be developed for the implant surgeon to facilitate proper implant placement based on the final prosthesis' occlusion and aesthetics.
Treatment planning software can also be used to demonstrate "try-ins" to the patient on a computer screen. When options have been fully discussed between patient and surgeon, the same software can be used to produce precision drill guides. Specialized software applications such as 'SimPlant' (simulated implant) or 'NobelGuide' use the digital data from a patient's CBCT to build a treatment plan. A data set is then produced and sent to a lab for production of a precision in-mouth drilling guide.[38]
Success rates Dental implant success is related to operator skill, quality and quantity of the bone available at the site, and the patient's oral hygiene. The consensus is that implants carry a success rate of around 75%[39]
One of the most important factors that determine implant success is the achievement and maintenance of implant stability.[40] The stability is presented as an ISQ (Implant Stability Quotient) value. Other contributing factors to the success of dental implant placement, as with most surgical procedures, include the patient's overall general health and compliance with post-surgical care.
Dental implants can help stop periodontal diseases from forming. This is because gaps formed by missing teeth can cause periodontal disease and infections that may affect a person’s ability to eat or speak. It is very important to fill in any gaps to prevent the adjacent and opposing set of teeth from shifting positions or causing further damage to the mouth.[41]
Failure Failure of a dental implant is often related to the failure of the implant to osseointegrate correctly with the bone, or vice-versa. A dental implant is considered to be a failure if it is lost, mobile or shows peri-implant (around the implant) bone loss of greater than 1.0 mm in the first year and greater than 0.2 mm a year after.
Dental implants are not susceptible to dental caries but they can develop a condition called peri-implantitis. This is an inflammatory condition of the mucosa and/or bone around the implant which may result in bone loss and eventual loss of the implant. The condition is usually, but not always, associated with a chronic infection. Peri-implantitis is more likely to occur in heavy smokers, patients with diabetes, patients with poor oral hygiene and cases where the mucosa around the implant is thin.[42]
Currently there is no universal agreement on the best treatment for peri-implantitis. The condition and its causes is still poorly understood.[43]
Risk of failure is increased in smokers. For this reason implants are frequently placed only after a patient has stopped smoking as the treatment is very expensive. More rarely, an implant may fail because of poor positioning at the time of surgery, or may be overloaded initially causing failure to integrate. If smoking and positioning problems exist prior to implant surgery, clinicians often advise patients that a bridge or partial denture rather than an implant may be a better solution.
Failure may also occur independently of the causes outlined above. Implants like any other object suffers from wear and tear. If the implant(s)in question are replacing commonly used teeth, then these may suffer from wear and tear and after years may crack and break up, although this is a very rare occurrence.
In the majority of cases where an implant fails to integrate with the bone and is rejected by the body the cause is unknown. This may occur in around 5% of cases. To this day we still do not know why bone will integrate with titanium dental implants and why it does not reject the material as a 'foreign body'. Many theories have been postulated over the last five decades. A recent theory argues that rather than being an active biological tissue response, the integration of bone with an implant is the lack of a negative tissue response. In other words, for unknown reasons the usual response of the body to reject foreign objects implanted into it does not function correctly with titanium implants. It has further been postulated that an implant rejection occurs in patients whose bone tissues actually react as they naturally should with the 'foreign body' and reject the implant in the same manner that would occur with most other implanted materials.[44]
Contraindications There are few absolute contraindications to implant dentistry. However, there are some systemic, behavioral, and anatomic considerations that should be assessed.
Particularly for mandibular (lower jaw) implants, in the vicinity of the mental foramen (MF), there must be sufficient alveolar bone above the mandibular canal also called the inferior alveolar canal or IAC (which acts as the conduit for the neurovascular bundle carrying the inferior alveolar nerve or IAN).
Failure to precisely locate the IAN and MF invites surgical insult by the drills and the implant itself. Such insult may cause irreparable damage to the nerve, often felt as a paresthesia (numbness) or dysesthesia (painful numbness) of the gum, lip and chin. This condition may persist for life and may be accompanied by unconscious drooling.
Uncontrolled Type II diabetes is a significant relative contraindication as healing following any type of surgical procedure is delayed due to poor peripheral blood circulation. Anatomic considerations include the volume and height of bone available. Often an ancillary procedure known as a block graft or sinus augmentation are needed to provide enough bone for successful implant placement.
There is new information about intravenous and oral bisphosphonates (taken for certain forms of breast cancer and osteoporosis, respectively) which may put patients at a higher risk of developing a delayed healing syndrome called osteonecrosis. Implants are contraindicated for some patients who take intravenous bisphosphonates.
The many millions of patients who take an oral bisphosphonate (such as Actonel, Fosamax and Boniva) may sometimes be advised to stop the administration prior to implant surgery, then resume several months later. However, current evidence suggests that this protocol may not be necessary. As of January 2008, an oral bisphosphonate study reported in the February 2008 Journal of Oral and Maxillofacial Surgery, reviewing 115 cases that included 468 implants, concluded "There is no evidence of bisphosphonate-associated osteonecrosis of the jaw in any of the patients evaluated in the clinic and those contacted by phone or e-mail reported no symptoms."[45]
The American Dental Association had addressed bisphosphonates in an article entitled "Bisphosphonate medications and your oral health,"[46] In an Overview, the ADA stated "The risk of developing BON [bisphosphonate-associated osteonecrosis of the jaw] in patients on oral bisphosphonate therapy appears to be very low...". The ADA Council on Scientific Affairs also employed a panel of experts who issued recommendations [for clinicians] for treatment of patients on oral bisphosphonates, published in June 2006. The overview may be read online at ada.org but it has now been superseded by a huge study—encompassing over 700,000 cases—entitled "Bisphosphonate Use and the Risk of Adverse Jaw Outcomes." Like the 2008 JOMS study, the ADA study exonerates oral bisphosphonates as a contraindication to dental implants.[47]
Bruxism (tooth clenching or grinding) is another consideration which may reduce the prognosis for treatment. The forces generated during bruxism are particularly detrimental to implants while bone is healing; micromovements in the implant positioning are associated with increased rates of implant failure. Bruxism continues to pose a threat to implants throughout the life of the recipient.[48] Natural teeth contain a periodontal ligament allowing each tooth to move and absorb shock in response to vertical and horizontal forces. Once replaced by dental implants, this ligament is lost and teeth are immovably anchored directly into the jaw bone. This problem can be minimized by wearing a custom made mouthguard (such an NTI appliance) at night.
Postoperatively, after implants have been placed, there are physical contraindications that prompt rapid action by the implantology team. Excessive or severe pain lasting more than three days is a warning sign, as is excessive bleeding. Constant numbness of the gingiva (gum), lip and chin—usually noticed after surgical anesthesia wears off—is another warning sign. In the latter case, which may be accompanied by severe constant pain, the standard of care calls for diagnosis to determine if the surgical procedure insulted the IAN. A 3D cone beam X-ray provides the necessary data, but even before this step a prudent implantologist may back out or completely remove an implant in an effort to restore nerve function because delay is usually ineffective. Depending upon the evidence visible with a 3D X-ray, patients may be referred to a specialist in nerve repair. In all cases, speed in diagnosis and treatment are necessary.
Market In the United States and the United Kingdom, there is no exclusive specialty in 'implantology'.
Any practitioner who carries out implant treatment, whether in the surgical insertion or the final provision of the prosthesis, must be adequately trained. Legal training requirements differ between countries.
In 2008, in the UK the General Dental Council (GDC) laid down strict training requirements[49] for dentists involved in dental implantology. Any dentist in the UK who wishes to train in the field of dental implantology must take part in an extended learning program which covers a detailed theory syllabus, as approved by the GDC,[50] in addition to formal supervised surgical training and mentoring. Dentists must not take part in implant dentistry in the UK until they have been approved by the training provider as having passed a formal competency assessment. Failure to comply with the GDC regulations may result in a dentist being removed from the Dental Register and hence losing the right to practice dentistry in the UK.[51]
The fast growing demand for prosthetic solutions based upon dental implants caused the birth of hundreds of different brands available on the market, about 150 in the UK[52] and more than 300 in Italy,[53] but only a very few of these have enough documentation and clinical practice for being recognized worldwide.[53] According to a study published in the Clinical Implant Dentistry and Related Research, only three brands of implants were tested by at least two independent (not sponsored) clinical researches,[54] which should push the patients to ask for more information before accepting any treatment, and to demand for a real implant passport.
Orthodontics
Orthodontics, formerly orthodontia (from Greek orthos "straight or proper or perfect"; and odous "tooth") is the first specialty of dentistry that is concerned with the study and treatment of malocclusions (improper bites), which may be a result of tooth irregularity, disproportionate jaw relationships, or both. Orthodontic treatment can focus on dental displacement only, or can deal with the control and modification of facial growth. In the latter case it is better defined as "dentofacial orthopaedics". Orthodontic treatment can be carried out for purely aesthetic reasons with regards to improving the general appearance of patients' teeth. However, there are orthodontists who work on reconstructing the entire face rather than focusing exclusively on teeth.
Contents Modern Orthodontics The use of digital models in orthodontics is rapidly increasing as the industry undergoes analog to digital conversions in record keeping. The University of Minnesota recently developed Three Dimensional Dental Models for Computer Automated Treatment Simulation that can be used to reduce the amount of human input needed for orthodontic treatment planning. This software tool has the ability to automatically segment teeth from one another and the gums. Digital laboratories are currently being used by many orthodontists, but can be very expensive. This software provides an efficient and cost-effective method for completing the segmentation process.[1]
Methods For comprehensive orthodontic treatment, most commonly, metal wires (Juste) are inserted into orthodontic brackets (see dental braces), which can be made from stainless steel or a more aesthetic ceramic material. The wires interact with the brackets to move teeth into the desired positions. Other methods may include invisalign. Invisalign consists of clear plastic aligners that move teeth.
Dental braces, with a powerchain, removed after completion of treatment. Additional components—including removable appliances ("plates"), headgear, expansion appliances, and many other devices—may also be used to move teeth and jaw bones. Functional appliances, for example, are used in growing patients (age 5 to 14) with the aim of modifying the jaw dimensions and relationship if these are altered. This therapy, termed Dentofacial Orthopedics, is frequently followed by fixed multibracket therapy ("full braces") to align the teeth and refine the occlusion.
Hawley retainers are the most common type of retainers. This picture shows retainers for the top and bottom of the mouth. Orthodontics is the study of dentistry that is concerned with the treatment of improper bites, and crooked teeth. Orthodontic treatment can help fix your teeth and set them in the right place. Orthodontists usually use braces and retainers to set your teeth.[2] There are, however, orthodontists who work on reconstructing the entire face rather than focusing exclusively on teeth. After a course of active orthodontic treatment, patients will typically wear retainers, which maintain the teeth in their improved positions while surrounding bone reforms around them. The retainers are generally worn full-time for a period, anywhere from just a few days to a year, then part-time (typically, nightly during sleep) for as long as the orthodontist recommends. It is possible for the teeth to stay aligned without regular retainer wear. However, there are many reasons teeth will crowd as a person ages, whether or not the individual ever experienced orthodontic treatment; thus there is no guarantee that teeth will stay aligned without retention. For this reason, many orthodontists prescribe night-time or part-time retainer wear for many years after orthodontic treatment (potentially for life). Adult orthodontic patients are more likely to need lifetime retention.
Diagnosis and treatment planning In diagnosis and treatment planning, the orthodontist must (1) recognize the various characteristics of a malocclusion or dentofacial deformity; (2) define the nature of the problem, including the etiology if possible;(3) design a treatment strategy based on the specific needs and desires of the individual; and (4) present the treatment strategy to the patient in such a way that the patient fully understands the ramifications of his/her decision.[3]
The New York Times has recently written that orthodontists are using Cone Beam CT too much in the diagnosis and treatment of orthodontic patients, leading to an unnecessary increased risk of cancer.[4]
Training Orthodontics was the first recognized specialty field within dentistry. Many countries have their own systems for training and registering orthodontic specialists. A two- to three-year period of full-time post-graduate study is required for a dentist to qualify as an orthodontist.
United States of America In order to be enrolled as a student or a resident in Advanced Education in Orthodontics approved by the Commission on Dental Accreditation (CODA), the dentist must have graduated with a DDS, DMD, BDS or equivalent. In the U.S., the orthodontics residency duration lasts between 24 –36 months long. The orthodontic residency typically award Certificate (non-degree) or Master of Science degree depending on its research requirements. The class size, tuition, stipend and number of patients seen and treated will all depend on the location of the program (hospital vs. university). Each training program has its own goals and treatment philosophy, however, most U.S. orthodontic programs focus on fixed straight wire appliances. All the graduates must also complete the written portion of the American Board of Orthodontics (ABO) examinations.
In order to become a Board Certified orthodontist, the practicing orthodontist must present six cases that have been treated entirely by the orthodontist to the ABO examiners. Once certified, the certificate is renewed every 10 years and the practitioner can add the following title, Diplomate, American Board of Orthodontics.
Similar to the ABO, the Canadian orthodontic specialist can take a two-part examinations (Written NDSE and Oral NDSE) offered by the Royal College of Dentists of Canada (RCDC) in their final year of the orthodontics training. Upon completion of the examinations, the orthodontist is admitted to the RCDC as a Fellow and can add the following title, FRCD(C).
Job outlook as a recent U.S. orthodontic graduates depend on the location of employment. Typically, more popular destinations (such as California, NYC, Seattle, Las Vegas and Texas) are heavily saturated with orthodontists. Traditional, practice transition situations (new orthodontist buying out the seller orthodontist) are becoming rare as the orthodontic market has been saturated with new and old practitioners, general dentists performing orthodontics, corporate clinics and older orthodontists delaying retirement.
Europe In the United Kingdom, this training period lasts three years, after completion of a membership from a Royal College. A further two years is then completed to train to consultant level, after which a fellowship examination from the Royal College is sat. In other parts of Europe, a similar pattern is followed. It is always worth contacting the professional body responsible for registering orthodontists to ensure that the orthodontist you wish to consult is a recognised specialist.
Canada A number of dental schools and hospitals offer advanced education in the specialty of Orthodontics to dentists seeking postgraduate education. The courses range from two to three years (with the majority being 3 years) of full-time classes in the theoretical and practical aspects of orthodontics together with clinical experience. Generally, admission is based on an application process followed by an extensive interviewing process by the institution, in order to select the best candidates. Candidates usually have to contact the individual school directly for the application process.
India In India, many dental colleges affiliated to universities offer orthodontics as specialization in Master of Dental Surgery ( M.D.S ) programme.The minimum qualification for M.D.S is Bachelor of Dental Surgery ( B.D.S ). The present course for MDS in Orthodontics stands at 3 years in all dental colleges in India which are recognised by the Dental Council of India.
The Indian Orthodontic Society was established in 1965. The Academy of Fixed Orthodontics, (AFO),[5] established in 2004, represents GP's and members from other dental specialties (Non Orthodontists) who practices orthodontics. AFO offers certification courses in Fixed Orthodontics for General Practitioners in Dentistry.
Iran In Iran, Orthodontics and Dentofacial Orthopedics is known as a specialty since 1978 and general dentists after participating in a comprehensive national exam( which is held once a year) and fulfill good rankings among all participants, can begin their postgraduate course. After three years of postgraduate academic training, students participate in a national board exam for final evaluation and finally if they can reach the determined scores, they will be recognized as specialist orthodontists. Iranian Association of Orthodontists(IAO) is established in 1978 and has been contributed in several national and international congresses since its establishment.
Contents Modern Orthodontics The use of digital models in orthodontics is rapidly increasing as the industry undergoes analog to digital conversions in record keeping. The University of Minnesota recently developed Three Dimensional Dental Models for Computer Automated Treatment Simulation that can be used to reduce the amount of human input needed for orthodontic treatment planning. This software tool has the ability to automatically segment teeth from one another and the gums. Digital laboratories are currently being used by many orthodontists, but can be very expensive. This software provides an efficient and cost-effective method for completing the segmentation process.[1]
Methods For comprehensive orthodontic treatment, most commonly, metal wires (Juste) are inserted into orthodontic brackets (see dental braces), which can be made from stainless steel or a more aesthetic ceramic material. The wires interact with the brackets to move teeth into the desired positions. Other methods may include invisalign. Invisalign consists of clear plastic aligners that move teeth.
Dental braces, with a powerchain, removed after completion of treatment. Additional components—including removable appliances ("plates"), headgear, expansion appliances, and many other devices—may also be used to move teeth and jaw bones. Functional appliances, for example, are used in growing patients (age 5 to 14) with the aim of modifying the jaw dimensions and relationship if these are altered. This therapy, termed Dentofacial Orthopedics, is frequently followed by fixed multibracket therapy ("full braces") to align the teeth and refine the occlusion.
Hawley retainers are the most common type of retainers. This picture shows retainers for the top and bottom of the mouth. Orthodontics is the study of dentistry that is concerned with the treatment of improper bites, and crooked teeth. Orthodontic treatment can help fix your teeth and set them in the right place. Orthodontists usually use braces and retainers to set your teeth.[2] There are, however, orthodontists who work on reconstructing the entire face rather than focusing exclusively on teeth. After a course of active orthodontic treatment, patients will typically wear retainers, which maintain the teeth in their improved positions while surrounding bone reforms around them. The retainers are generally worn full-time for a period, anywhere from just a few days to a year, then part-time (typically, nightly during sleep) for as long as the orthodontist recommends. It is possible for the teeth to stay aligned without regular retainer wear. However, there are many reasons teeth will crowd as a person ages, whether or not the individual ever experienced orthodontic treatment; thus there is no guarantee that teeth will stay aligned without retention. For this reason, many orthodontists prescribe night-time or part-time retainer wear for many years after orthodontic treatment (potentially for life). Adult orthodontic patients are more likely to need lifetime retention.
Diagnosis and treatment planning In diagnosis and treatment planning, the orthodontist must (1) recognize the various characteristics of a malocclusion or dentofacial deformity; (2) define the nature of the problem, including the etiology if possible;(3) design a treatment strategy based on the specific needs and desires of the individual; and (4) present the treatment strategy to the patient in such a way that the patient fully understands the ramifications of his/her decision.[3]
The New York Times has recently written that orthodontists are using Cone Beam CT too much in the diagnosis and treatment of orthodontic patients, leading to an unnecessary increased risk of cancer.[4]
Training Orthodontics was the first recognized specialty field within dentistry. Many countries have their own systems for training and registering orthodontic specialists. A two- to three-year period of full-time post-graduate study is required for a dentist to qualify as an orthodontist.
United States of America In order to be enrolled as a student or a resident in Advanced Education in Orthodontics approved by the Commission on Dental Accreditation (CODA), the dentist must have graduated with a DDS, DMD, BDS or equivalent. In the U.S., the orthodontics residency duration lasts between 24 –36 months long. The orthodontic residency typically award Certificate (non-degree) or Master of Science degree depending on its research requirements. The class size, tuition, stipend and number of patients seen and treated will all depend on the location of the program (hospital vs. university). Each training program has its own goals and treatment philosophy, however, most U.S. orthodontic programs focus on fixed straight wire appliances. All the graduates must also complete the written portion of the American Board of Orthodontics (ABO) examinations.
In order to become a Board Certified orthodontist, the practicing orthodontist must present six cases that have been treated entirely by the orthodontist to the ABO examiners. Once certified, the certificate is renewed every 10 years and the practitioner can add the following title, Diplomate, American Board of Orthodontics.
Similar to the ABO, the Canadian orthodontic specialist can take a two-part examinations (Written NDSE and Oral NDSE) offered by the Royal College of Dentists of Canada (RCDC) in their final year of the orthodontics training. Upon completion of the examinations, the orthodontist is admitted to the RCDC as a Fellow and can add the following title, FRCD(C).
Job outlook as a recent U.S. orthodontic graduates depend on the location of employment. Typically, more popular destinations (such as California, NYC, Seattle, Las Vegas and Texas) are heavily saturated with orthodontists. Traditional, practice transition situations (new orthodontist buying out the seller orthodontist) are becoming rare as the orthodontic market has been saturated with new and old practitioners, general dentists performing orthodontics, corporate clinics and older orthodontists delaying retirement.
Europe In the United Kingdom, this training period lasts three years, after completion of a membership from a Royal College. A further two years is then completed to train to consultant level, after which a fellowship examination from the Royal College is sat. In other parts of Europe, a similar pattern is followed. It is always worth contacting the professional body responsible for registering orthodontists to ensure that the orthodontist you wish to consult is a recognised specialist.
Canada A number of dental schools and hospitals offer advanced education in the specialty of Orthodontics to dentists seeking postgraduate education. The courses range from two to three years (with the majority being 3 years) of full-time classes in the theoretical and practical aspects of orthodontics together with clinical experience. Generally, admission is based on an application process followed by an extensive interviewing process by the institution, in order to select the best candidates. Candidates usually have to contact the individual school directly for the application process.
India In India, many dental colleges affiliated to universities offer orthodontics as specialization in Master of Dental Surgery ( M.D.S ) programme.The minimum qualification for M.D.S is Bachelor of Dental Surgery ( B.D.S ). The present course for MDS in Orthodontics stands at 3 years in all dental colleges in India which are recognised by the Dental Council of India.
The Indian Orthodontic Society was established in 1965. The Academy of Fixed Orthodontics, (AFO),[5] established in 2004, represents GP's and members from other dental specialties (Non Orthodontists) who practices orthodontics. AFO offers certification courses in Fixed Orthodontics for General Practitioners in Dentistry.
Iran In Iran, Orthodontics and Dentofacial Orthopedics is known as a specialty since 1978 and general dentists after participating in a comprehensive national exam( which is held once a year) and fulfill good rankings among all participants, can begin their postgraduate course. After three years of postgraduate academic training, students participate in a national board exam for final evaluation and finally if they can reach the determined scores, they will be recognized as specialist orthodontists. Iranian Association of Orthodontists(IAO) is established in 1978 and has been contributed in several national and international congresses since its establishment.
CONTACT DETAIL
Clinic name: Al habib dental clinic
Clinic address: Aligarh bazar orangi town sector 2e karachi pakistan
City/province: karachi/sindh
mobile number: 0333-2158490
contact person: Dr Abdul Aziz Habib
show room: http/alhabibdentalclinic.weebly.com
Clinic address: Aligarh bazar orangi town sector 2e karachi pakistan
City/province: karachi/sindh
mobile number: 0333-2158490
contact person: Dr Abdul Aziz Habib
show room: http/alhabibdentalclinic.weebly.com
